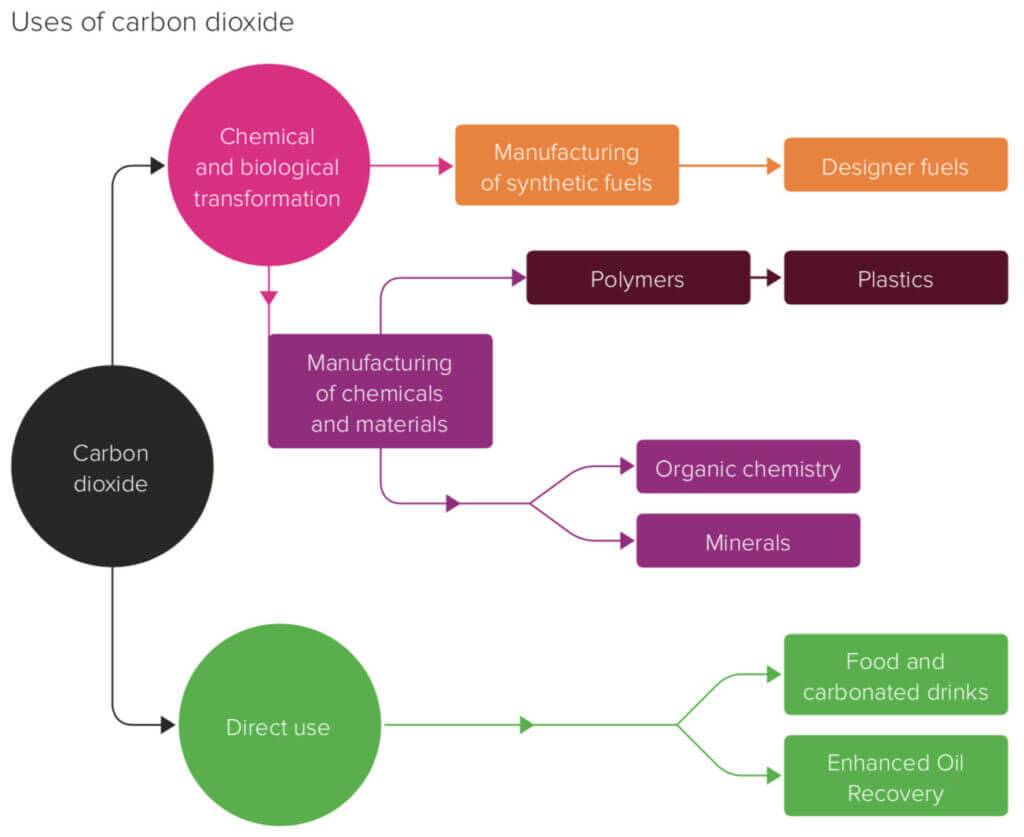
Contents
- Understanding the Differences Between Liquid and Gaseous CO2 in Industrial Applications
- The Properties of Liquid CO2
- Applications of Liquid CO2
- The Properties of Gaseous CO2
- Applications of Gaseous CO2
- Key Takeaways: Differences between Liquid and Gaseous CO2 in Industrial Applications
- Frequently Asked Questions
- What is the state of CO2 in its liquid and gaseous forms?
- What are the key differences in physical properties between liquid and gaseous CO2?
- How are liquid and gaseous CO2 used in industrial applications?
- What are the safety considerations for handling liquid and gaseous CO2?
- Are there any environmental considerations associated with liquid and gaseous CO2?
- Carbon Dioxide, liquid CO2, solid CO2 and gaseous CO2
- Final Thoughts
When it comes to industrial applications, understanding the differences between liquid and gaseous CO2 is crucial. So, what sets these two forms of carbon dioxide apart? Let’s dive in and explore the distinctions between liquid and gaseous CO2 in industrial settings.
In industrial applications, the state of CO2, whether it’s in liquid or gaseous form, plays a significant role in determining its usability and efficacy. Liquid CO2 is commonly used in various industries, such as food and beverage, pharmaceutical, and chemical manufacturing. This form of CO2 is created by compressing and cooling gaseous carbon dioxide, resulting in a dense liquid that is easier to handle and store. On the other hand, gaseous CO2 is the naturally occurring state of carbon dioxide when it exists at standard temperature and pressure. It is often used in applications where a gas is required, such as in fire extinguishers or as a propellant in aerosol products. Understanding the differences between these two states of CO2 is essential for selecting the appropriate form for specific industrial processes.
By exploring the unique characteristics of liquid and gaseous CO2, we can gain a deeper understanding of their applications in various industries. Whether it’s the convenience and versatility of liquid CO2 or the practicality of gaseous CO2, each form has its own advantages and uses. So, let’s delve further into the specifics and uncover the nuances between liquid and gaseous CO2 in industrial applications.

Understanding the Differences Between Liquid and Gaseous CO2 in Industrial Applications
Carbon dioxide (CO2) is a versatile compound that finds numerous applications in various industries. It can exist in two different states: liquid and gaseous. While both forms have their uses, understanding the differences between them is crucial for determining their suitability for specific industrial applications.
The Properties of Liquid CO2
Liquid CO2 is formed when carbon dioxide is subjected to low temperatures and high pressures. It is commonly stored in pressurized containers or tanks, where it remains in a liquid state. One of the key characteristics of liquid CO2 is its high density, which allows for the storage of large quantities in a relatively small space. This makes it an ideal choice for industries that require bulk storage of CO2.
In addition to its high density, liquid CO2 also exhibits a unique property known as the triple point. At this point, the temperature and pressure conditions are such that the substance can exist simultaneously as a solid, liquid, and gas. This property makes liquid CO2 particularly useful in applications where precise control of temperature and pressure is required, such as in food processing and pharmaceutical manufacturing.
Applications of Liquid CO2
Liquid CO2 has a wide range of industrial applications, thanks to its unique properties. One of its primary uses is as a solvent in extraction processes, where it is employed to extract valuable compounds from raw materials. For example, in the coffee industry, liquid CO2 is used to extract caffeine from coffee beans while preserving the flavor and aroma. Similarly, it is utilized in the extraction of essential oils from plants.
Another significant application of liquid CO2 is in the field of cryogenics. Due to its low temperature, it can be used as a cooling agent in various industrial processes. For instance, it is employed in the transportation and storage of frozen food, as well as in the preservation of biological samples and medical supplies.
Furthermore, liquid CO2 finds use in the manufacturing of carbonated beverages. When released from a pressurized container, the sudden drop in pressure causes the liquid CO2 to expand rapidly into gas, creating the characteristic fizziness in carbonated drinks.
The Properties of Gaseous CO2
Gaseous CO2, on the other hand, is the form of carbon dioxide that we encounter in our daily lives. It is a colorless and odorless gas that is naturally present in the Earth’s atmosphere. Gaseous CO2 is produced by various natural and human activities, including respiration, combustion, and industrial processes.
One of the defining properties of gaseous CO2 is its low density. In its gaseous state, CO2 molecules are more spread out and less tightly packed compared to the liquid form. This makes gaseous CO2 highly suitable for applications where dispersal is required, such as in fire suppression systems or as a coolant in electronic devices.
Applications of Gaseous CO2
Gaseous CO2 has a wide range of industrial applications due to its abundance and versatility. One of its primary uses is as a refrigerant in cooling systems. It is often used as a replacement for ozone-depleting refrigerants in air conditioning units and refrigeration systems. Gaseous CO2 is also utilized in welding and cutting processes, where it provides an inert atmosphere that prevents oxidation.
Additionally, gaseous CO2 is a crucial component in the production of carbonated beverages. When injected into a liquid, it dissolves and creates the characteristic bubbles in soft drinks and sparkling water. It is also utilized in the oil and gas industry for enhanced oil recovery, where it is injected into oil wells to increase pressure and facilitate the extraction of oil.
In conclusion, understanding the differences between liquid and gaseous CO2 is essential for selecting the appropriate form for specific industrial applications. Liquid CO2 is characterized by its high density and is often used in extraction processes, cryogenics, and the manufacturing of carbonated beverages. On the other hand, gaseous CO2 is known for its low density and finds applications in cooling systems, welding, and the production of carbonated beverages. Both forms have their unique properties and advantages, making them valuable resources in the industrial sector.
Key Takeaways: Differences between Liquid and Gaseous CO2 in Industrial Applications
- Liquid CO2 is stored under pressure and can be easily transported in tanks, while gaseous CO2 is generated on-site using a generator.
- Liquid CO2 is commonly used for cryogenic applications, such as freezing and cooling, while gaseous CO2 is often used for inerting and fire suppression.
- Liquid CO2 has a higher density and requires less storage space compared to gaseous CO2.
- Gaseous CO2 is easier to handle and distribute as it doesn’t require special storage and handling equipment like tanks.
- Both liquid and gaseous CO2 have various industrial applications, including food and beverage processing, pharmaceutical manufacturing, and wastewater treatment.
Frequently Asked Questions
Curious about the differences between liquid and gaseous CO2 in industrial applications? Look no further! We’ve got the answers to your questions right here.
What is the state of CO2 in its liquid and gaseous forms?
In its liquid form, CO2 is stored under high pressure and maintained at a temperature below -56.6 degrees Celsius (-69.88 degrees Fahrenheit). This results in the formation of a dense liquid that is commonly used for various industrial processes. On the other hand, gaseous CO2 is at atmospheric pressure and exists at temperatures above -56.6 degrees Celsius. It is the most common form of CO2 found in the atmosphere.
Both liquid and gaseous CO2 have distinct properties and are used in different industrial applications based on their specific characteristics.
What are the key differences in physical properties between liquid and gaseous CO2?
One of the main differences between liquid and gaseous CO2 lies in their density. Liquid CO2 is much denser than its gaseous counterpart, making it more suitable for storage and transportation in industrial settings. Additionally, liquid CO2 can be easily pumped and manipulated due to its high density.
Gaseous CO2, on the other hand, is lighter and less dense, which allows it to disperse more quickly in the air. This property makes gaseous CO2 ideal for applications such as carbonation, where it is used to add fizz to beverages or create a pressurized environment in certain manufacturing processes.
How are liquid and gaseous CO2 used in industrial applications?
Liquid CO2 finds extensive use in various industrial applications. It is commonly employed as a cooling agent, particularly in cryogenic processes where low temperatures are crucial. Liquid CO2 is also utilized as a solvent in extraction processes and as a raw material in the production of chemicals, such as urea and methanol.
Gaseous CO2, on the other hand, is widely used in the food and beverage industry for carbonation, as well as in the oil and gas industry for enhanced oil recovery. It is also utilized in fire suppression systems and as a shielding gas in welding operations.
What are the safety considerations for handling liquid and gaseous CO2?
Due to its extremely low temperature, liquid CO2 requires careful handling to prevent frostbite or thermal burns. Protective equipment, such as insulated gloves and goggles, should be worn when working with liquid CO2 to ensure safety. Additionally, proper ventilation is crucial when handling gaseous CO2 to prevent the build-up of high concentrations, which can be harmful to human health.
It is important to follow industry-specific safety guidelines and regulations when working with both liquid and gaseous CO2 to minimize the risk of accidents or exposure to hazardous conditions.
Are there any environmental considerations associated with liquid and gaseous CO2?
Both liquid and gaseous CO2 have environmental implications, particularly in relation to carbon emissions and climate change. CO2 is a greenhouse gas that contributes to global warming when released into the atmosphere. Therefore, it is important for industries to implement sustainable practices and minimize CO2 emissions.
However, it’s worth noting that gaseous CO2 can be captured and stored underground through carbon capture and storage (CCS) technologies, which helps mitigate its impact on the environment. The use of liquid CO2 in various industrial processes can also contribute to reducing carbon emissions when compared to alternative methods.
Carbon Dioxide, liquid CO2, solid CO2 and gaseous CO2
Final Thoughts
Now that we’ve explored the differences between liquid and gaseous CO2 in industrial applications, it’s clear that both forms have their own unique advantages and uses. Liquid CO2, with its high density and ability to be easily transported and stored, is ideal for applications that require large quantities of CO2, such as carbonation in the beverage industry or as a cooling agent in refrigeration systems. On the other hand, gaseous CO2 offers flexibility and convenience, making it suitable for applications like welding, fire suppression, and chemical processing.
When it comes to safety, it’s important to note that both liquid and gaseous CO2 can be hazardous if mishandled. Proper safety precautions should always be taken when working with either form. Additionally, the cost and availability of each form may vary depending on the specific industry and location.
In conclusion, understanding the differences between liquid and gaseous CO2 allows industries to make informed choices about which form is best suited for their specific needs. Whether it’s the convenience and flexibility of gaseous CO2 or the density and transportability of liquid CO2, both forms play a vital role in various industrial applications. So, next time you enjoy a carbonated beverage or benefit from a fire suppression system, you can appreciate the role that CO2, in its different states, plays in making it all possible.